Document Details
Document Information
86e223ba-dcdc-44b5-a7cf-8a6b0bf40bf8
Biogen to Highlight New Lecanemab Data and Scientific Advances at the 18th Clinical Trials on Alzhei
press_release
Company Communication Type CEO Executives CEO
None
2025-11-20
N/A
2233
30583
Actions
Document Content
# Biogen to Highlight New Lecanemab Data and Scientific Advances at the 18th Clinical Trials on Alzheimer's Disease Conference **Date:** 2025-11-20 07:30:00 **Company:** Biogen Inc. **Ticker:** BIIB **Source URL:** https://www.globenewswire.com/news-release/2025/11/20/3191792/0/en/Biogen-to-Highlight-New-Lecanemab-Data-and-Scientific-Advances-at-the-18th-Clinical-Trials-on-Alzheimer-s-Disease-Conference.html --- [Accessibility: Skip TopNav](https://www.globenewswire.com/news-release/2025/11/20/3191792/0/en/Biogen-to-Highlight-New-Lecanemab-Data-and-Scientific-Advances-at-the-18th-Clinical-Trials-on-Alzheimer-s-Disease-Conference.html#maincontainer) [](https://www.globenewswire.com/) - [Newsroom](https://www.globenewswire.com/newsroom) - [Services](https://www.globenewswire.com/services) - [Contact Us](https://insight.notified.com/globenewswire-contact-us?utm_medium=Website&utm_source=Contact%20...
# Biogen to Highlight New Lecanemab Data and Scientific Advances at the 18th Clinical Trials on Alzheimer's Disease Conference **Date:** 2025-11-20 07:30:00 **Company:** Biogen Inc. **Ticker:** BIIB **Source URL:** https://www.globenewswire.com/news-release/2025/11/20/3191792/0/en/Biogen-to-Highlight-New-Lecanemab-Data-and-Scientific-Advances-at-the-18th-Clinical-Trials-on-Alzheimer-s-Disease-Conference.html --- [Accessibility: Skip TopNav](https://www.globenewswire.com/news-release/2025/11/20/3191792/0/en/Biogen-to-Highlight-New-Lecanemab-Data-and-Scientific-Advances-at-the-18th-Clinical-Trials-on-Alzheimer-s-Disease-Conference.html#maincontainer) [](https://www.globenewswire.com/) - [Newsroom](https://www.globenewswire.com/newsroom) - [Services](https://www.globenewswire.com/services) - [Contact Us](https://insight.notified.com/globenewswire-contact-us?utm_medium=Website&utm_source=Contact%20Us&utm_campaign=Contact%20Us%20ENG) - [About Us](https://www.globenewswire.com/about) - English [Sign In](https://www.globenewswire.com/home/signin) [Register](https://portal.notified.com/en/registration) # [](https://www.biogen.com/) # Biogen to Highlight New Lecanemab Data and Scientific Advances at the 18th Clinical Trials on Alzheimer’s Disease Conference November 20, 2025 07:30 ET\| Source: [Biogen Inc.](https://www.globenewswire.com/en/search/organization/Biogen%2520Inc%C2%A7)FollowBiogen Inc. * * * Share - * * * - New lecanemab findings highlight the safety and potential benefits of subcutaneous administration for initiation dosing, the potential for additional long-term clinical benefits with continued treatment, and real-world experience - Biogen is committed to deepening scientific understanding of Alzheimer’s disease, including therapeutic delivery and disease progression CAMBRIDGE, Mass., Nov. 20, 2025 (GLOBE NEWSWIRE) -- [Biogen](https://www.globenewswire.com/Tracker?data=RndSyw6ZZJ_Mv1JJ_g8MB5r2EFE-7IWNp8aDrU9ePeEfhDPvsFz8JO6QEt_lDQIF00zC3TnaExgBACPyeYQ-W0QXkrzWoA4sCGh7Dy3w3xA2DvTwqptI0Db_1SmdSOjRdgn35DVnerzKCteRBOLdF-HXuNQoDF11E-k1_S1OUS8dRKveo6cRj1QSIg3h3R0zYmSX0naNISWfyKjWF9Anpj3zlgLrE549vpLpuRRR1nE= "Biogen") Inc. (Nasdaq: BIIB) today announced upcoming scientific presentations at the 18th Clinical Trials on Alzheimer’s Disease (CTAD) Conference, taking place December 1-4 in San Diego. Data on LEQEMBI® (lecanemab-irmb) will include findings on subcutaneous administration for initiation dosing, the benefits of continued therapy and estimated time savings over 10 years of treatment based on Phase 3 clinical data, and real-world experience from a post-marketing observational study in Japan and the ALZ-NET registry. The company will also share new insights into BIIB080, an investigational antisense oligonucleotide (ASO) therapy, from a healthy-volunteer biodistribution study, as well as research that helps inform understanding of disease progression. “Following the introduction of LEQEMBI IQLIK™ in the U.S. for maintenance therapy, we look forward to sharing data that explore its potential for initiation dosing and how it may expand treatment options for patients and care partners across both phases of the treatment journey,” said Priya Singhal, M.D., M.P.H., Head of Development at Biogen. “The breadth of data at CTAD also underscores the growing evidence supporting the long-term benefits of continued treatment with LEQEMBI and reflects our ongoing commitment to advancing scientific understanding of Alzheimer’s disease.” **Key Scientific Sessions and Presentations** **Late-Breaking Symposium:** - “Lecanemab Subcutaneous Formulation for Treatment Initiation in Early Alzheimer’s Disease: Optimizing Patient Care with a Potential New Option,” on Wednesday, December 3 from 3:10-3:50 p.m. PT **Oral Presentations**: - “The Effects of Lecanemab Treatment on Soluble CSF Aβ Protofibrils in Clarity AD,” on Tuesday, December 2 from 1:40-1:55 p.m. PT - “Benefit Continues to Accumulate When Treatment is Continued Beyond Plaque Clearance Estimating Accumulated or Maintained Treatment Benefit in the CLARITY AD and TRAILBLAZER-ALZ2 Trials,” on Tuesday, December 2 from 5:05--5:20 p.m. PT - “Estimating the 10-Year Time-Savings Benefits of Lecanemab Treatment,” on Wednesday, December 3 from 2:40-2:55 p.m. PT - “Interim Analysis of Post-Marketing Observational Study of Lecanemab in Japan,” on Thursday, December 4 from 11:40-11:55 a.m. PT **Poster Presentations:** - “Baseline Characteristics and Preliminary Safety from a Multicenter, Safety Surveillance Study of Lecanemab Treatment for Alzheimer’s Disease in Real-World Clinical Practice” on Tuesday, December 2 from 7:15 a.m.-5:30 p.m. PT - “Real-World Clinical Safety and Patient-Reported Outcomes of Treatment with Lecanemab in a New England Alzheimer’s Disease Center” on Tuesday, December 2 from 7:15 a.m.-5:30 p.m. PT - “Stability and Improvement in Early Alzheimer’s Disease with Lecanemab: Sub-analysis from a United States Multicenter, Retrospective, Real-World Study” on Tuesday, December 2 from 7:15 a.m.-5:30 p.m. PT - “Biodistribution of Radiolabeled MAPT Antisense Oligonucleotide BIIB080 Following Intrathecal Administration in Healthy Adults,” on Wednesday, December 3 from 7:15 a.m.-5:30 p.m. PT - “Baseline Study-Partner Reported but not Participant Self-Reported ECog-12 score is Associated with Subsequent Cognitive Progression on CDR-SB over 78 Weeks of Follow-up,” on Thursday, December 4 from 7:15 a.m. -5:00 p.m. PT **About LEQEMBI** **®** **(lecanemab-irmb)** LEQEMBI (lecanemab-irmb) is the result of a strategic research alliance between Eisai and BioArctic. LEQEMBI is a humanized immunoglobulin gamma 1 (IgG1) monoclonal antibody directed against aggregated soluble (protofibril) and insoluble forms of amyloid-beta (Aβ). LEQEMBI is an amyloid beta-directed antibody for the treatment for Alzheimer’s disease (AD) in the U.S. The U.S. Food and Drug Administration (FDA) granted LEQEMBI traditional approval on July 6, 2023. LEQEMBI is indicated for the treatment of Alzheimer’s disease. Treatment with LEQEMBI should be initiated in patients with mild cognitive impairment or mild dementia stage of disease, the population in which treatment was initiated in clinical trials. Eisai and Biogen have been collaborating on the joint development and commercialization of AD treatments since 2014. Eisai serves as the lead of LEQEMBI development and regulatory submissions globally with both companies co-commercializing and co-promoting the product and Eisai having final decision-making authority. Please see full U.S. [Prescribing Information](https://www.globenewswire.com/Tracker?data=wxfy30zQs_A1qQIzGYbIQchG8WSWuge60Q7JaRta5Hu1-vpk4NkrKa81fVmA9GmHwsTYhAMednWrRhMHJPLfdCzGYq1QOCqY-PoZqV9Yabis6ijWf4hqVct7moY3NwdNNir69kQLvtG8FfFjY2RcBWAuSYfmUmdprX_T5UqxjyyQ7A4A8S4JLK8dF3NfjvX9lwO_q0kUBLXqczNdM9DJs1t4vL36H6FMJMjz2WRsW4QVWG1R0hjv7P52CtjtzDDSTtcTwi8YzHf_-LQEG9cM636PNakuDiDeUGjFm9w71rHI2gsTjrhA3IXSVOdFibKSb0UUwG8xPiB5b6oUguFr2Puq5Pwc3Fd32SWtRno7YNHJLogoJzU1MLfRVI7PvH198ePFZOlix4YLq91mpxEgdWwKMzWFnlaC3xiqlsGOzIwbNxsr5ob4rJYOlNZ8wnkzX3UJ0m-_RZejN9B9lqQeh5Dx22i94I85xlpNs8jbJy1MxCN-5XwMtmMGa0I8721WdjLVYqgmagSgr3L5JJ3mboZ0wS4Twx5taPqt6Vg41yrvVb8I4FqhwRuRQNRKgsWpcFCZf3vkUBxDGn9gsHjX2aWvkj2QSSmxLXueO0yg-pfAS_LsZBZBJENE3QbCk8wOHp-2Ggkkac-ySDzByUPQmVF_To4JpScfG6XWpILKBuwwKjQjVAhGIiwKSKklo-eI3IUv2aTeuup4W54Q2r019w== "Prescribing Information") for LEQEMBI, including Boxed WARNING and [Medication Guide](https://www.globenewswire.com/Tracker?data=wQQLK8M9acWtJyPhzA0BqyMLc7_6Mh5ZheSRn8ajZMoOSFXbxed2Mh7_0-LBVpNKqfCpC6xtbtEE9IkwOhPxCHjlUsNKt-MdcpkbhSJmemudmN1kg_rk-4OdpBAAcCvfBXzLBDO0PxmDSZfnxkJkMHqT1Ohq2PcyoDDgqbW4WyHuGLA5YQ1vKJ5MeU7Kc1oIc-iPlG3XSQQKu6gsb-J3WvH1rK17kHkP0QpI7pGOrIrWgOiEwmRuqut0VOI8KVSZ0KBWO95qLsLlmp0SHWuB9bg4AoZm_wG6fPUW5YbGtp1rSEL28ZWYd3xq3QUeXNkUa-SYb8mXr7oSc9dGoEGAYBe2Evuqo5WAar_4NuPI4iyXqki_gv3emu3m8ei-QuKQhpJSqIPodaHW9IbLpTzOLa2pfUt8FI7vs94alak4cIydSUDud80N_uTdxpMvYcLS5JjWnXgUqNQ-rnD0hcmugZ8w2sTkAYl70ra7frkDoQYkZby5zaY1MIsONf9kdpNShOba0xeSBVLJhOUn3GEZx8H7LJQnV0Ua6jfFKiZzUIu6UQpBn9rbqNku_vWm5PBaUZczI6jcwmDr8b7ek1dekdtodD9GyGaeGExBzoqy2FaaVFwwCXmE4jTadN82DAJVIN3UlbEHJc8yVwzYTeYepw== "Medication Guide"). **About BIIB080** BIIB080 is an investigational antisense oligonucleotide (ASO) therapy designed to target microtubule-associated protein tau (MAPT) mRNA to reduce the production of tau protein. Abnormal accumulation of tau in the brain is a hallmark of Alzheimer’s disease and is associated with neurodegeneration and cognitive decline. BIIB080 is currently being evaluated in a Phase 2 clinical study (NCT05399888) in individuals with early Alzheimer’s disease. In December 2019, Biogen exercised a license option with Ionis Pharmaceuticals and obtained a worldwide, exclusive, royalty-bearing license to develop and commercialize BIIB080 (tau ASO). **About Biogen** Founded in 1978, Biogen is a leading biotechnology company that pioneers innovative science to deliver new medicines to transform patient’s lives and to create value for shareholders and our communities. We apply deep understanding of human biology and leverage different modalities to advance first-in-class treatments or therapies that deliver superior outcomes. Our approach is to take bold risks, balanced with return on investment to deliver long-term growth. We routinely post information that may be important to investors on our website at [www.biogen.com](https://www.globenewswire.com/Tracker?data=IVb0ECXWRkuNUZYohnAKCM5MhHdEh9CKUWkB3c0tKUsPe7niUzyKH7_wuUKL5wpijNIjvB43XUX2G1uXxgNB4DCPF1UN7Cod-5XP73lIlmqONALwTUjkaHM-pGRMhE194U-No7a9y-qaUu3cvJ32pkyF4w-DnHXpp3E2YFN_raSwm4iJj0Lgh_RaqpnpkrLZ55KFhd-8nxOWTJlgVT19QUT0pwxyxLh1WeIApD6lKdI= "www.biogen.com"). Follow Biogen on social media – [Facebook](https://www.globenewswire.com/Tracker?data=orquZq5pwJTe31CF0Hp8IjJHOdG5WZgK1AKtctlaclpCtfiuYa5l7QUvEHTY9c_BTrMORR0zdvv08qdnf7QjIx7UqCc6-IuRjmKS0rklwJvCy23haDWDtNInW3djlZOxD2OtOM8dVGmaWzQXZnUX_NcnBvMCWUEdfN22WZh6k3QWgvEvr4QFa6I73kjj3y6BAe7MfJA26XwDBjsCV5FTONS26ie4KA6Uk71y5_KjZuuuyNVN9QTQ6P6nOI624BHlOhHljazHlCsbvQ778oTUQ9pQ4qloXJf3GBAo5nVd-rDWNpCopKMAVSK7GfVGd4pT7K12IRK_H6mQYJHuYAqCJCaJBg0ZBFqbsR1UmFVeRRjA8ABxMy8EA5ZlnrPiOei_mIhcL445YMRgfzYoJ3w1jrPSvVtB4VscqrrPFx-D14kZK_7v-ZAzaK1Z5Wq9pXYH39l3zk6HZA8_5Zfb3d1Uh3tHwMIByEFOg5zE5uflOI1nmyS4sqimwJq7KMfwRj95wxWQAGRqbdCoQ0lgkn_Qksfmg1DWnQaTbupZg77s6i4HjStAoBTqEKhoZtBd6hqQ "Facebook"), [LinkedIn](https://www.globenewswire.com/Tracker?data=v5pV6ntLMK-PdKTBpc61kDGBcN8DVASclHxBjpLTCcNCdI40KtovVhEFBsPKAHP1qb6qHyXm7GUNidN497zIPfF4aT1Y6GpErNhc417UgX-oIGzbI32GhO1lTutIQhLiwDZY_P7FP7WS0PEPLVT-Lt0z7TNAGCiPDMmBQviSweTmXm5ksxU1v25F_Chnu0JU7E_y7uJDZ2tvKJGonoxXmPYFH4H7_ib0Fw7DzIw8aFZXIKoo1H5twXT_ZnR4BsSjbM0EC8auk7QsStLX088F4oF3fCrMVbXZgPWjgutVvTBfFASLRO3ziRbAsULrElVR2MPKo6FEE9vhsCztmlHBhXZrOlkCHoBGfyY8ptdtkEs8DAEyKsPqE0jePA4BaWTR4T41Pv2xWfi6az4gEFoZEMVooB1nGScW8WUCL5L6s4INpGi_42Rcrb76Ggm3chdHuU17NiGv7U9xw_5lGFlydrnhD7TLV8csNLGFGGKGJzHSuZtPm0hY2upcY824y7UFBdcdAKU2llmTtZXSRvgb64GuzsT_Ldz5LAkTYXYO-WEwykHjsSxxpmPk4ySbn26e "LinkedIn"), [X](https://www.globenewswire.com/Tracker?data=dHyhsUGltLg86CaQra7LfemhMVqFSkdLxrnj18sWWhQoP1ppC1Wa5gJvxE1x6GY0pkGS01tU5iFdVAk8s44BvwFBA6wE2CHPGtQKlMGkSDPpTMfIm72AkpAwGpC3UmNctwHioQpE-k5g6DDeXNhC8NdQGhVDCFhlQHi6dSoGte2z8GPCxG43OOTDnC3Faw9mXocvBCLBBwUCaucf1EfdSQ== "X"), [YouTube](https://www.globenewswire.com/Tracker?data=Wqgz67cTNugiMctYk2Lpsv3K3RpSPJPmuneZS_j97RARaa9DNx0HNNbAXRO7NeXrfcErTlljghiqzgjZ2eGn8H_WfeBIvxEVqNtP0gsEXj9qbUJYfJSBXFlDW9MDqaBn_tEeKN9GbYaKixt3h5OvyOtLf8CExNHmDA-uOxxsIzB4w1T29idRWzEgRiLUgSX7_wKl8GS8MPee6pt_9eMyhxlWI1-AGSL1zBHDlg_FMLZ0wAXId-84szrkUGBdn0XFyO_o_EsP3D-nnhkwyEzmIxkFdClMVw9qoP93w0LzFupfMjibdcEDE_GOWTEM4AazNXewtDQN3eDgx4LvjboCRGxFF1hQ44tmS0XpmsvKvVaIry5q5ZeC3Ac0XE3x2ZazwRg7jxhjOieTG0s6pbCXqspnxfs0zwL1n6-leVmPuJgKVo1hzLC2_-VhTF5CIXiTfvEjK4vCUx0KOoDJ7k3UHQeYP6TXi1rtNdPFCM_He4-qNAqFgSBqGJ13M-8CUAcu1isBMGplKsoRNWceJTTlzF41ocytAUzss9aFk3wsI1YBHwvFGqDQCCWq5SaEYMGU "YouTube"). **Biogen Safe Harbor** This news release contains forward-looking statements, including about the potential clinical effects of lecanemab; the potential benefits, safety and efficacy of lecanemab ; potential regulatory discussions, submissions and approvals and the timing thereof; the treatment of Alzheimer's disease; the anticipated benefits and potential of Biogen's collaboration arrangements with Eisai; the potential of Biogen's commercial business and pipeline programs, including lecanemab; and risks and uncertainties associated with drug development and commercialization. These forward-looking statements may be accompanied by such words as “aim,” “anticipate,” “believe,” “contemplate,” “continue,” “could,” “estimate,” “expect,” "forecast," “goal,” “guidance”, “hope,” ”intend,” “may,” “objective,” “plan,” “possible,” “potential,” “predict,” “project,” “prospect,” “should,” “target,” “will,” “would”: and other words and terms of similar meaning. Drug development and commercialization involve a high degree of risk, and only a small number of research and development programs result in commercialization of a product. Results in early-stage clinical trials may not be indicative of full results or results from later stage or larger scale clinical trials and do not ensure regulatory approval. You should not place undue reliance on these statements. Given their forward-looking nature, these statements involve substantial risks and uncertainties that may be based on inaccurate assumptions and could cause actual results to differ materially from those reflected in such statements. These forward-looking statements are based on management's current beliefs and assumptions and on information currently available to management. Given their nature, we cannot assure that any outcome expressed in these forward-looking statements will be realized in whole or in part. We caution that these statements are subject to risks and uncertainties, many of which are outside of our control and could cause future events or results to be materially different from those stated or implied in this document, including, among others, uncertainty of long-term success in developing, licensing, or acquiring other product candidates or additional indications for existing products; expectations, plans and prospects relating to product approvals, approvals of additional indications for our existing products, sales, pricing, growth, reimbursement and launch of our marketed and pipeline products; our ability to effectively implement our corporate strategy; the successful execution of our strategic and growth initiatives, including acquisitions; the risk that positive results in a clinical trial may not be replicated in subsequent or confirmatory trials or success in early stage clinical trials may not be predictive of results in later stage or large scale clinical trials or trials in other potential indications; risks associated with clinical trials, including our ability to adequately manage clinical activities, unexpected concerns that may arise from additional data or analysis obtained during clinical trials, regulatory authorities may require additional information or further studies, or may fail to approve or may delay approval of our drug candidates; the occurrence of adverse safety events, restrictions on use with our products, or product liability claims; and any other risks and uncertainties that are described in other reports we have filed with the U.S. Securities and Exchange Commission. These statements speak only as of the date of this press release and are based on information and estimates available to us at this time. Should known or unknown risks or uncertainties materialize or should underlying assumptions prove inaccurate, actual results could vary materially from past results and those anticipated, estimated or projected. Investors are cautioned not to put undue reliance on forward-looking statements. A further list and description of risks, uncertainties and other matters can be found in our Annual Report on Form 10-K for the fiscal year ended December 31, 2024 and in our subsequent reports on Form 10-Q and Form 10-K, in each case including in the sections thereof captioned “Note Regarding Forward-Looking Statements” and “Item 1A. Risk Factors,” and in our subsequent reports on Form 8-K. Except as required by law, we do not undertake any obligation to publicly update any forward-looking statements whether as a result of any new information, future events, changed circumstances or otherwise. **Digital Media Disclosures** From time to time, we have used, or expect in the future to use, our investor relations website (investors.biogen.com), the Biogen LinkedIn account (linkedin.com/company/biogen) and the Biogen X account ( [https://x.com/biogen](https://www.globenewswire.com/Tracker?data=7UhbsZ70wluYWKbZ9bSyC_OifE2SW0w2pNvB5qTkRunODtyg8q2aocMYvE4t_uHL3Ma_lJeLMCTr6OcITlnaeRazgDZOrLOQnCVxqWFGSNqNl3zO9Eu55C1WkZNAtiKgC14b96Rf3rIZtMK9OFpyqtUxdNzlLvi2D5Thi2qzbbo4s6eDSFinTrqHXyJAgwEda60O12ZdOTPp9D3lvjKkIE3pE51bZ5iOYZcmhMhqRaOWz9QCMomcUrZxNgxqgNntXqJrpgETNjpeyTdCtLVH6g== "")) as a means of disclosing information to the public in a broad, non-exclusionary manner, including for purposes of the SEC’s Regulation Fair Disclosure (Reg FD). Accordingly, investors should monitor our investor relations website and these social media channels in addition to our press releases, SEC filings, public conference calls and websites, as the information posted on them could be material to investors. | | | | --- | --- | | MEDIA CONTACT:<br>**Biogen**<br>Madeleine Shin<br>\+ 1 781 464 3260<br>[public.affairs@biogen.com](mailto:public.affairs@biogen.com "public.affairs@biogen.com") | INVESTOR CONTACT:<br>**Biogen**<br>Tim Power<br>+1 781 464 2442<br>[IR@biogen.com](mailto:IR@biogen.com "IR@biogen.com") |  GlobeNewswire Company ProfileBiogen Inc.Industry: BiotechnologyWebsite: [https://www.biogen.com/](https://www.biogen.com/) Press Release Actions - [Print](https://www.globenewswire.com/news-release/2025/11/20/3191792/0/en/Biogen-to-Highlight-New-Lecanemab-Data-and-Scientific-Advances-at-the-18th-Clinical-Trials-on-Alzheimer-s-Disease-Conference.html?print=1) - [Download PDF](https://www.globenewswire.com/news-release/2025/11/20/3191792/0/en/Biogen-to-Highlight-New-Lecanemab-Data-and-Scientific-Advances-at-the-18th-Clinical-Trials-on-Alzheimer-s-Disease-Conference.html?pdf=1) - [Subscribe via RSS](https://www.globenewswire.com/rssfeed/organization/hNQxyS-FvexsgfoXWT62ZA==) - [Subscribe via ATOM](https://www.globenewswire.com/atomfeed/organization/hNQxyS-FvexsgfoXWT62ZA==) - [Javascript](https://www.globenewswire.com/JSWidget/organization/JtSgc9eWhzTeChxZhHYxww%3d%3d) ## Recommended Reading - March 31, 2026 06:59 ET\|Source: [Biogen Inc.](https://www.globenewswire.com/en/search/organization/Biogen%2520Inc%C2%A7) [Biogen to Acquire Apellis, Enhancing the Company’s Growth Portfolio in Immunology and Rare Disease, Bolstering Growth Outlook and Accelerating Expansion into Nephrology](https://www.globenewswire.com/news-release/2026/03/31/3265288/0/en/Biogen-to-Acquire-Apellis-Enhancing-the-Company-s-Growth-Portfolio-in-Immunology-and-Rare-Disease-Bolstering-Growth-Outlook-and-Accelerating-Expansion-into-Nephrology.html) Acquisition will bring two differentiated commercialized immunology medicines to Biogen with EMPAVELI® FDA-approved in three indications, including two rare kidney diseases, and SYFOVRE® FDA-approved... [Read More](https://www.globenewswire.com/news-release/2026/03/31/3265288/0/en/Biogen-to-Acquire-Apellis-Enhancing-the-Company-s-Growth-Portfolio-in-Immunology-and-Rare-Disease-Bolstering-Growth-Outlook-and-Accelerating-Expansion-into-Nephrology.html)  - March 30, 2026 07:30 ET\|Source: [Biogen Inc.](https://www.globenewswire.com/en/search/organization/Biogen%2520Inc%C2%A7) [FDA Approves New High Dose Regimen of SPINRAZA® (nusinersen) for Spinal Muscular Atrophy](https://www.globenewswire.com/news-release/2026/03/30/3264519/0/en/FDA-Approves-New-High-Dose-Regimen-of-SPINRAZA-nusinersen-for-Spinal-Muscular-Atrophy.html) The approval was anchored on data from the pivotal DEVOTE study that investigated the efficacy and safety of the High Dose Regimen of SPINRAZA in treatment-naïve and previously treated SPINRAZA... [Read More](https://www.globenewswire.com/news-release/2026/03/30/3264519/0/en/FDA-Approves-New-High-Dose-Regimen-of-SPINRAZA-nusinersen-for-Spinal-Muscular-Atrophy.html)  [View More News](https://www.globenewswire.com/en/newsroom) ## Explore Previous [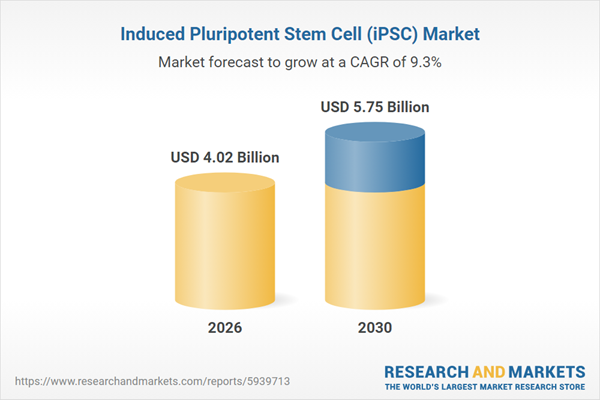](https://www.globenewswire.com/news-release/2026/04/14/3273620/0/en/Trends-and-Strategies-Shaping-the-4-Billion-Induced-Pluripotent-Stem-Cell-iPSC-Market-2026-2030-Major-Collaborations-and-Advances-in-iPSC-Technology-Unveiled.html) April 14, 2026 10:35 ET [Trends and Strategies Shaping the $4+ Billion Indu...](https://www.globenewswire.com/news-release/2026/04/14/3273620/0/en/Trends-and-Strategies-Shaping-the-4-Billion-Induced-Pluripotent-Stem-Cell-iPSC-Market-2026-2030-Major-Collaborations-and-Advances-in-iPSC-Technology-Unveiled.html "Trends and...") [](https://www.globenewswire.com/news-release/2026/04/14/3273618/0/en/Trust-and-Corporate-Services-Industry-to-Grow-by-3-Billion-Over-2026-2030-Vistra-Group-Intertrust-Group-and-TMF-Group-Lead.html) April 14, 2026 10:32 ET [Trust and Corporate Services Industry to Grow by $...](https://www.globenewswire.com/news-release/2026/04/14/3273618/0/en/Trust-and-Corporate-Services-Industry-to-Grow-by-3-Billion-Over-2026-2030-Vistra-Group-Intertrust-Group-and-TMF-Group-Lead.html "Trust and Corporate...") [](https://www.globenewswire.com/news-release/2026/04/14/3273616/0/en/CBIZ-Elevates-Peter-Scavuzzo-to-Oversee-Unified-Technology-Organization.html) April 14, 2026 10:30 ET [CBIZ Elevates Peter Scavuzzo to Oversee Unified Te...](https://www.globenewswire.com/news-release/2026/04/14/3273616/0/en/CBIZ-Elevates-Peter-Scavuzzo-to-Oversee-Unified-Technology-Organization.html "CBIZ Elevates Peter...") [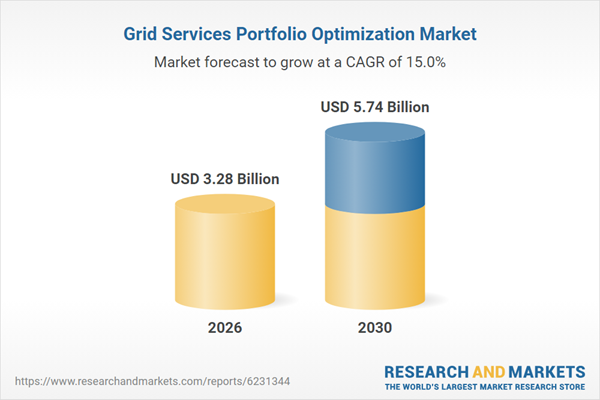](https://www.globenewswire.com/news-release/2026/04/14/3273622/0/en/Grid-Services-Portfolio-Optimization-Market-Report-2026-with-5-Years-Historic-Analysis-and-a-10-Year-Growth-Forecast.html) April 14, 2026 10:38 ET [Grid Services Portfolio Optimization Market Report...](https://www.globenewswire.com/news-release/2026/04/14/3273622/0/en/Grid-Services-Portfolio-Optimization-Market-Report-2026-with-5-Years-Historic-Analysis-and-a-10-Year-Growth-Forecast.html "Grid Services...") [](https://www.globenewswire.com/news-release/2026/04/14/3273621/0/en/The-Best-Stage-During-Coachella-2026-Wasn-t-on-the-Map.html) April 14, 2026 10:37 ET [The Best Stage During Coachella 2026 Wasn't on the...](https://www.globenewswire.com/news-release/2026/04/14/3273621/0/en/The-Best-Stage-During-Coachella-2026-Wasn-t-on-the-Map.html "The Best Stage...") [](https://www.globenewswire.com/news-release/2026/04/14/3273620/0/en/Trends-and-Strategies-Shaping-the-4-Billion-Induced-Pluripotent-Stem-Cell-iPSC-Market-2026-2030-Major-Collaborations-and-Advances-in-iPSC-Technology-Unveiled.html) April 14, 2026 10:35 ET [Trends and Strategies Shaping the $4+ Billion Indu...](https://www.globenewswire.com/news-release/2026/04/14/3273620/0/en/Trends-and-Strategies-Shaping-the-4-Billion-Induced-Pluripotent-Stem-Cell-iPSC-Market-2026-2030-Major-Collaborations-and-Advances-in-iPSC-Technology-Unveiled.html "Trends and...") [](https://www.globenewswire.com/news-release/2026/04/14/3273618/0/en/Trust-and-Corporate-Services-Industry-to-Grow-by-3-Billion-Over-2026-2030-Vistra-Group-Intertrust-Group-and-TMF-Group-Lead.html) April 14, 2026 10:32 ET [Trust and Corporate Services Industry to Grow by $...](https://www.globenewswire.com/news-release/2026/04/14/3273618/0/en/Trust-and-Corporate-Services-Industry-to-Grow-by-3-Billion-Over-2026-2030-Vistra-Group-Intertrust-Group-and-TMF-Group-Lead.html "Trust and Corporate...") [](https://www.globenewswire.com/news-release/2026/04/14/3273616/0/en/CBIZ-Elevates-Peter-Scavuzzo-to-Oversee-Unified-Technology-Organization.html) April 14, 2026 10:30 ET [CBIZ Elevates Peter Scavuzzo to Oversee Unified Te...](https://www.globenewswire.com/news-release/2026/04/14/3273616/0/en/CBIZ-Elevates-Peter-Scavuzzo-to-Oversee-Unified-Technology-Organization.html "CBIZ Elevates Peter...") [](https://www.globenewswire.com/news-release/2026/04/14/3273622/0/en/Grid-Services-Portfolio-Optimization-Market-Report-2026-with-5-Years-Historic-Analysis-and-a-10-Year-Growth-Forecast.html) April 14, 2026 10:38 ET [Grid Services Portfolio Optimization Market Report...](https://www.globenewswire.com/news-release/2026/04/14/3273622/0/en/Grid-Services-Portfolio-Optimization-Market-Report-2026-with-5-Years-Historic-Analysis-and-a-10-Year-Growth-Forecast.html "Grid Services...") [](https://www.globenewswire.com/news-release/2026/04/14/3273621/0/en/The-Best-Stage-During-Coachella-2026-Wasn-t-on-the-Map.html) April 14, 2026 10:37 ET [The Best Stage During Coachella 2026 Wasn't on the...](https://www.globenewswire.com/news-release/2026/04/14/3273621/0/en/The-Best-Stage-During-Coachella-2026-Wasn-t-on-the-Map.html "The Best Stage...") [](https://www.globenewswire.com/news-release/2026/04/14/3273620/0/en/Trends-and-Strategies-Shaping-the-4-Billion-Induced-Pluripotent-Stem-Cell-iPSC-Market-2026-2030-Major-Collaborations-and-Advances-in-iPSC-Technology-Unveiled.html) April 14, 2026 10:35 ET [Trends and Strategies Shaping the $4+ Billion Indu...](https://www.globenewswire.com/news-release/2026/04/14/3273620/0/en/Trends-and-Strategies-Shaping-the-4-Billion-Induced-Pluripotent-Stem-Cell-iPSC-Market-2026-2030-Major-Collaborations-and-Advances-in-iPSC-Technology-Unveiled.html "Trends and...") [](https://www.globenewswire.com/news-release/2026/04/14/3273618/0/en/Trust-and-Corporate-Services-Industry-to-Grow-by-3-Billion-Over-2026-2030-Vistra-Group-Intertrust-Group-and-TMF-Group-Lead.html) April 14, 2026 10:32 ET [Trust and Corporate Services Industry to Grow by $...](https://www.globenewswire.com/news-release/2026/04/14/3273618/0/en/Trust-and-Corporate-Services-Industry-to-Grow-by-3-Billion-Over-2026-2030-Vistra-Group-Intertrust-Group-and-TMF-Group-Lead.html "Trust and Corporate...") [](https://www.globenewswire.com/news-release/2026/04/14/3273616/0/en/CBIZ-Elevates-Peter-Scavuzzo-to-Oversee-Unified-Technology-Organization.html) April 14, 2026 10:30 ET [CBIZ Elevates Peter Scavuzzo to Oversee Unified Te...](https://www.globenewswire.com/news-release/2026/04/14/3273616/0/en/CBIZ-Elevates-Peter-Scavuzzo-to-Oversee-Unified-Technology-Organization.html "CBIZ Elevates Peter...") Next About Us GlobeNewswire is one of the world's largest newswire distribution networks, specializing in the delivery of corporate press releases, financial disclosures and multimedia content to media, investors, and consumers worldwide. Global News - [English](https://www.globenewswire.com/newsroom) - [Français](https://www.globenewswire.com/fr/newsroom) - [Deutsch](https://www.globenewswire.com/de/newsroom) Newswire Distribution Network & Management - [Home](https://www.globenewswire.com/) - [Newsroom](https://www.globenewswire.com/newsroom) - [RSS Feeds](https://www.globenewswire.com/rss/list) - [Notified](https://www.notified.com/) - [Legal](https://www.notified.com/privacy) - [About Us](https://www.globenewswire.com/about) - [Contact Us](https://insight.notified.com/globenewswire-contact-us?utm_medium=Website&utm_source=Contact%20Us&utm_campaign=Contact%20Us%20ENG) - [Resources](https://www.notified.com/resources) © 2026 Digital Media Innovations, LLC. All rights reserved. [GlobeNewswire Linkedin](https://www.linkedin.com/company/1006947 "GlobeNewswire Linkedin") [Notified Twitter](https://twitter.com/Notified "Notified Twitter") ; [Français](https://www.globenewswire.com/fr/news-release/2025/11/20/3191792/0/en/Biogen-to-Highlight-New-Lecanemab-Data-and-Scientific-Advances-at-the-18th-Clinical-Trials-on-Alzheimer-s-Disease-Conference.html) [Deutsch](https://www.globenewswire.com/de/news-release/2025/11/20/3191792/0/en/Biogen-to-Highlight-New-Lecanemab-Data-and-Scientific-Advances-at-the-18th-Clinical-Trials-on-Alzheimer-s-Disease-Conference.html)